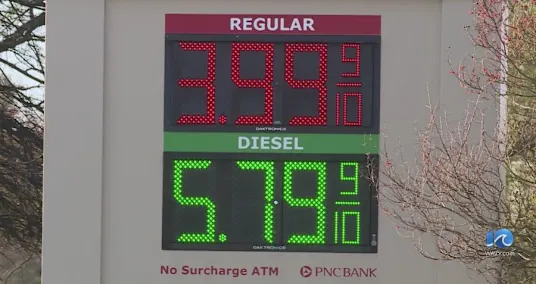(Reuters) -The U.S. Food and Drug Administration is probing the death of a patient who developed harmful antibodies after taking Takeda Pharmaceuticals' blood disorder therapy, the health regulator said on Friday.
The pediatric patient died about 10 months after starting Takeda's drug Adzynma as a preventive therapy, the agency said.
The child had congenital thrombotic thrombocytopenic purpura (cTTP), an inherited condition that causes blood clots in small vessels and can lead to organ damage.
The FDA said the child developed antibodies that blocked the activity of ADAMTS13, an enzyme critical for blood clotting.
Takeda did not immediately respond to Reuters request for comment.
Adzynma, approved in 2023 as the first therapy for cTTP, replaces the ADAMTS13 protein to help prevent dangerous blood clots.
The agency added it has received multiple postmarketing reports of patients developing neutralizing antibodies to ADAMTS13 after treatment with Adzynma.
(Reporting by Kamal Choudhury in Bengaluru; Editing by Vijay Kishore)
LATEST POSTS
- 1
 This Huge Ocean Beast Shifts Sharks’ Evolutionary Timeline
This Huge Ocean Beast Shifts Sharks’ Evolutionary Timeline - 2
 Manual for Vegetarian Protein Powder
Manual for Vegetarian Protein Powder - 3
 Our 10 favorite Space.com reader astronomy photos of 2025
Our 10 favorite Space.com reader astronomy photos of 2025 - 4
 2024 Moving Styles for Kitchen Redesigns
2024 Moving Styles for Kitchen Redesigns - 5
 From a new flagship space telescope to lunar exploration, global cooperation – and competition – will make 2026 an exciting year for space
From a new flagship space telescope to lunar exploration, global cooperation – and competition – will make 2026 an exciting year for space
 Will Comet C/2025 R3 (PanSTARRS) be the 'great comet' of 2026?
Will Comet C/2025 R3 (PanSTARRS) be the 'great comet' of 2026? Pain at the pump for Hampton Roads residents
Pain at the pump for Hampton Roads residents The most effective method to Decisively Use Open Record Rewards
The most effective method to Decisively Use Open Record Rewards Elite Execution Wall televisions for Film Darlings
Elite Execution Wall televisions for Film Darlings Outside Lovers' Decision: Favored Climbing Rucksacks
Outside Lovers' Decision: Favored Climbing Rucksacks The Best Games Crossroads in History
The Best Games Crossroads in History Nutrient Rich Natural products: Lift Your Wellbeing
Nutrient Rich Natural products: Lift Your Wellbeing Portugal among EU countries with the most people working close to 50 hours a week
Portugal among EU countries with the most people working close to 50 hours a week Old photos misrepresented as aftermath of political party supporters' brawl in Bangladesh
Old photos misrepresented as aftermath of political party supporters' brawl in Bangladesh